A sweet spot on JAGGED1 important for Notch signal generation
A new study published in Science Signalling has revealed an important role for a sugar attached to JAGGED1 in activating a core cellular developmental signalling pathway in humans. Professor Penny Handford's team, in collaboration with the groups of Dr Weston Struwe and Professor Mark Sansom at the University of Oxford, and Professor Cecilia Sahlgren at the University of Turku, have revealed that sugar modification of a membrane-interacting face of ligand JAGGED1 (see Figure 1) is important for activating the Notch receptor, most likely through positioning the ligand/receptor complex at the cell membrane.
It is well known that sugar modifications of Notch play a key role in its regulation, but they have not previously been implicated in optimising ligand function.
The sugar studied here was attached to a specific region of JAGGED1 (known as the C2 domain) that is implicated in binding to cell surfaces.
The scientists initially screened a cancer database to identify potential functionally important sites in JAGGED1, and then used mutagenesis procedures to intentionally alter key residues in the C2 domain that affect the attachment of the sugar, before observing the effects of these changes on different types of cells.
Additional procedures and simulations were then performed to suggest how the presence or absence of the sugar on the C2 domain might affect JAGGED1 binding to the surface of cells, key for Notch activation.
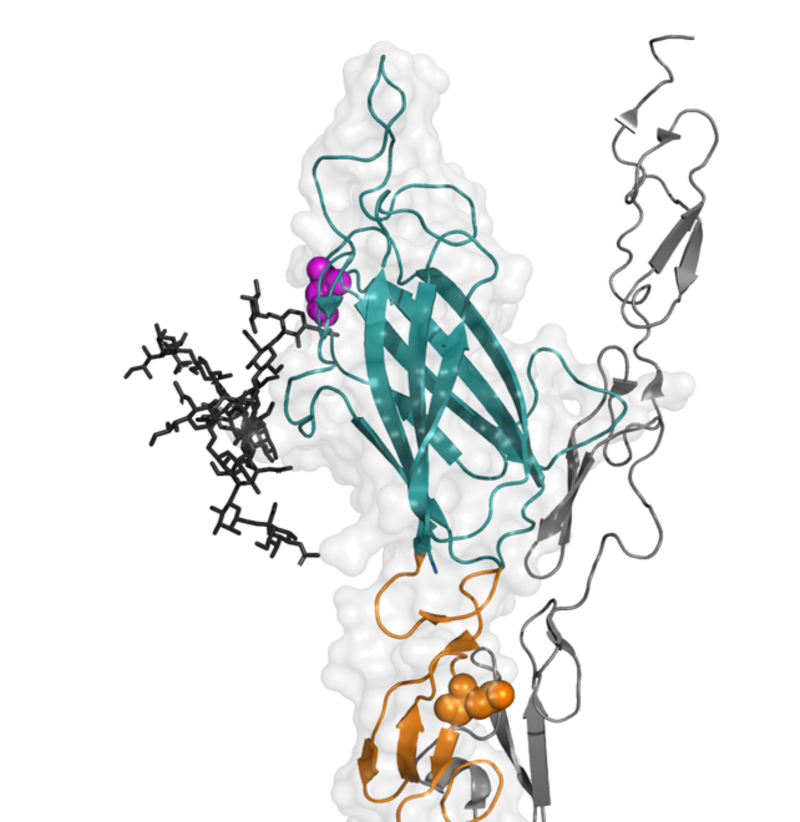
Figure 1: An N-glycan on JAGGED1 is important for Notch activation. Structure of JAGGED1 with the N-glycan shown attached to a residue (PINK) on the membrane-binding loop of the C2 domain ( TEAL), based on homologous structures. Notch ligand-binding r
Professor Handford from the University of Oxford's Department of Biochemistry explains: "The spotlight on the role of the ligand sugar came about through identifying an interesting mutation present in the COSMIC cancer database which was associated with unexplained loss of JAGGED1 function. This led to identification of a specific sugar type (known as an N-glycan) which has a key role in influencing how JAGGED1 binds to the membrane to optimise Notch signal generation."
In normal Notch signalling, Jagged-1 binds to a Notch receptor which transduces a signal into the cell through proteolysis, resulting in the regulation of gene expression in the nucleus. When this pathway is disrupted, many inherited and acquired diseases can arise including CADASIL, Alagille syndrome, as well as various cancers. These new findings have shown the importance of the correct positioning of the JAGGED1/Notch complex at the cell membrane, thereby increasing knowledge of the precise mechanism by which signal generation is achieved, important for future research.

Professor Penny Handford
This work was supported by the Medical Research Council as well as the InFLAMES Flagship Programme of the Academy of Finland, the Carlsberg Foundation, and funding from a UK Research and Innovation (UKRI) Future Leaders Fellowship.
Publication: An N-glycan on the C2domain of JAGGED1 is important for Notch activation
DOI: 10.1126/scisignal.abo3507




