Many anticancer drugs result in severe side effects due to damage inflicted on healthy cells. An important goal in drug development is to target drugs that will only kill cancerous cells. Folate is an essential vitamin (B9) obtained via our diet and is required for cell growth. Antifolate drugs kill cancerous cells but also damage healthy cells resulting in challenging side effects for patients. A new study from the Newstead group, published in Nature, now reveals the structure of the folate transporter, laying the foundation for developing targeted chemotherapy agents.
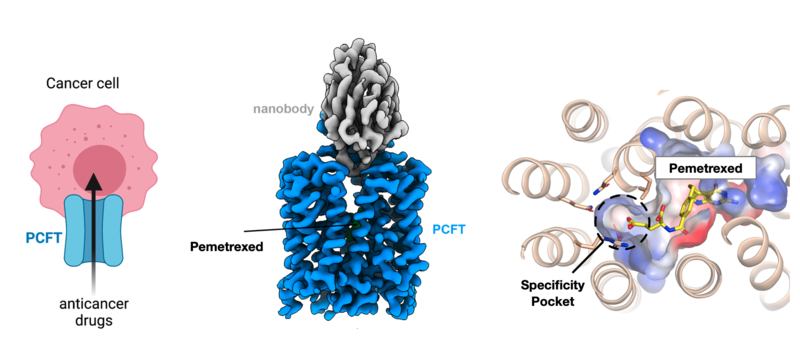
PCFT plays an important role in targeting chemotherapy drugs to cancer cells. The cryo-EM structure of PCFT now enables a more rational approach to anticancer drug design
Folate is transported into our cells through the proton coupled folate transporter (PCFT). PCFT transports dietary folates across the intestinal tract and into the brain. Importantly, this transporter is more active in cancerous cells due to their altered metabolism, offering a route to selectively target diseased tissue. However, current antifolate drugs are also recognised by other folate transport systems required by healthy cells to survive. The key is to target only cancerous cells by selective transport via PCFT.
In the latest research from the Newstead group and undertaken by Dr Jo Parker and aided by Dr Gabriel Kuteyi in collaboration with Dr Justin Deme & Prof Susan Lea in the Dunn School of Pathology but recently relocated to the National Cancer Institute at the National Institutes of Health in the USA and Dr Zhiyi Wu & Prof. Philip Biggin in the Department of Biochemistry now reveals the structure of PCFT for the first time.
Crucially, PCFT was solved in complex with Pemetrexed, a chemotherapy agent used to treat non-small cell lung cancer and pleural mesothelioma. These new results now reveal the blueprint to modify antifolate drugs to target PCFT more effectively.
Reference:
Simon Newstead
25th May 2021